Life Sciences Industrial Strategy & Sector Deal Newsletter

Life Sciences Industrial Strategy & Sector Deal Newsletter
Welcome to the first Life Sciences Industrial Strategy & Sector Deal Newsletter. Here you will find an update on how we are progressing with the implementation of phase 1 of the Life Sciences Sector Deal as well as our upcoming priorities and engagement opportunities.
Delivery of the Life Sciences Industrial Strategy and Sector Deal: Progress Highlights
The Sector Deal built on the ambitious Life Sciences Industrial Strategy, which set out recommendations for our world-leading life sciences industry to drive growth, increase productivity, improve the use of data, reinforce our science base, deepen our skills and secure benefits for patients throughout the UK.
Since the Sector Deal was launched on the 6th December 2017, we have made considerable progress.
Together with the sector Government has:
Ideas:
- Commissioned a £30m Vanguard Study, the first phase of our work to ultimately whole-genome-sequence all 500,000 participants of UK Biobank, which will provide many opportunities for the industry to benefit from a better understanding of disease.
- Worked with industry and other stakeholders to fully scope the £50m competition for a digital pathology and radiology programme with AI which launched on 6 June and closed on 2nd August (see below for further details).
Business Environment:
- Set up the Accelerated Access Collaborative (AAC) which is aiming to launch the Accelerated Access Pathway later this year, and appointed Lord Darzi as the chair of the AAC.
- Awarded £130m in support for industrial medicines manufacturing from the Industrial Strategy Challenge Fund so far, across a range of projects.
- Announced the Medicines Manufacturing Innovation Centre will be based in Scotland, a £56m project that includes £13m Industrial Strategy Challenge Fund support. The new centre will help companies develop processes and technologies for manufacturing medicines, reducing the time it takes for new medicines to reach the market.
- Awarded £21m ISCF funding to establish three Advanced Therapies Treatment Centres, in partnership with the NHS and industry, which will transform the UK’s ability to develop and deliver cell and gene therapies to patients
- Health Data Research UK (HDR UK), the national institute for health data science led by Professor Andrew Morris, will be leading delivery of the Digital Innovation Hubs (DIHs) and has announced a £37.5m investment. HDR UK will engage with a wide range of industry sectors to support delivery of the DIHs, including technology, wearables/devices, pharmaceutical and biotechnology companies.
- £2.5bn British Patient Capital programme launched to enable long-term investment in innovative companies across the UK, part of the Government’s response to the Patient Capital Review and alongside the private sector will leverage £7.5bn in more patient capital to help innovation-led businesses start and grow. You can read more about it here.
People
- Launched SIP Cambridge, a new science and industry skills partnership which looks to address the region’s skills gap and which will pave the way for collaborative approaches to tackling skills challenges.
- Launched the SIP apprenticeship survey (now closed), which gave industry and other sector organisations a chance to inform government analysis of apprenticeship uptake across the sector, enabling skills gaps in priority subjects to be addressed.
The governance framework, which oversees and supports the implementation of the Sector Deal, is also now in place and we’ve had successful first meetings earlier this year of both the Life Sciences Council and Life Sciences Industrial Strategy Implementation Board.
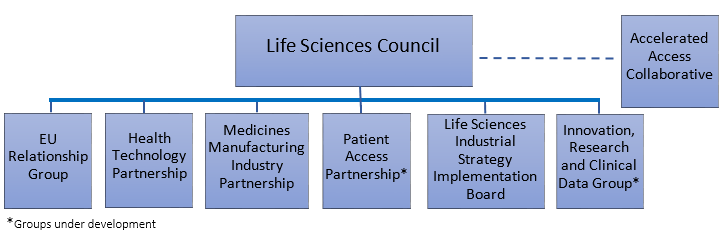
- The Life Sciences Council, chaired by Greg Clark and Jeremy Hunt, met for the first time on 16th May 2018 at Number 10; read more about what happened here.
- The Life Sciences Industrial Strategy Implementation Board (LSISIB), co-chaired by Sir Professor John Bell and Lord Henley, met for a second time on 20th June 2018 to review progress on the Sector Deal commitments and progress the discussion from the Life Sciences Council on priority areas to focus on next to implement the Strategy’s vision.
- The review process for the Expressions of Interest for ISCF Wave 3 is underway: 252 EoIs for ISCF Wave 3 were received by the 18th April closing date and the shortlist of proposed challenges is currently being considered. The next stage will refine and adapt those initial proposals to form the final list of compelling challenges. As part of that process, some potential challenges, although not all, will be holding open engagement events to refine the process. The final challenges will be announced in due course
Both Government and industry partners have been committed to maintaining the momentum and this progress has only been possible thanks to key partners working closely together.
ISCF Wave 2 Update
- ISCF competition: data to early diagnosis and precision medicine (launched 6th June 2018)
- As part of the Life Sciences Sector Deal and the Early Diagnosis Mission, we are supporting the creation of 5 to 6 centres of excellence across the UK, in digital pathology and/or medical imaging, including radiology. The centres must use digital systems and artificial intelligence (AI) to improve diagnosis and deliver precision treatments.
- Innovate UK, as part of UK Research and Innovation (UKRI), are investing up to £50 million to establish the centres of excellence as part of the delivery of the Industrial Strategy Challenge Fund (ISCF) in data to early diagnosis and precision medicine.
- The ISCF competition launched on 6th June 2018 and closed on 2nd August 2018. Please click here for further details.
Engaging on Future Policy Development
We want to ensure that the progress we have already made so far continues, and are always keen to hear your ideas and/or proposals and how we can incorporate these into the next phase of work to implement the Life Sciences Industrial Strategy.
Areas being considered by the Life Sciences Industrial Strategy Implementation Board and the Life Sciences Council for further work, with potential to feed into the next phase of the Life Sciences Sector Deal, include:
- Early diagnostics: further work on developing the concept and supporting programmes for a Health Advanced Research Programme (which was first conceptualised in the Life Sciences Industrial Strategy published last Summer);
- Clinical trials: improving the UK’s clinical trial capabilities, making the UK the best destination for conducting clinical trials and leveraging the UK and NHS data and digital capabilities;
- Innovative regulation: working to take the lead in this area to ensure that regulation supports innovation in the life sciences sector, for example in emerging technologies such as AI algorithms, cell and gene therapies, and digital medicines;
- Skills: ensuring the sector’s skills agenda is aligned to the Life Sciences Industrial Strategy and creates economic growth with the right skills base for the future and for employers;
- Growing UK clusters: developing growth of key clusters and providing a single gateway for investors;
- AI in health: including AI applications in drug discovery, medical research and robotic surgery.
The areas outlined above are in early stages of development and packages are not yet finalised. If you have a proposal that you are looking to take forward, we recommend linking in with the expert groups, as outlined above, via the trade associations in the first instance.
Other News from OLS
We recently responded to the Lords Science and Technology Committee report on the Life Sciences Industrial Strategy. The response was published on 27th June 2018 on the Parliament website and can be read in full here.
.png)
.png)